 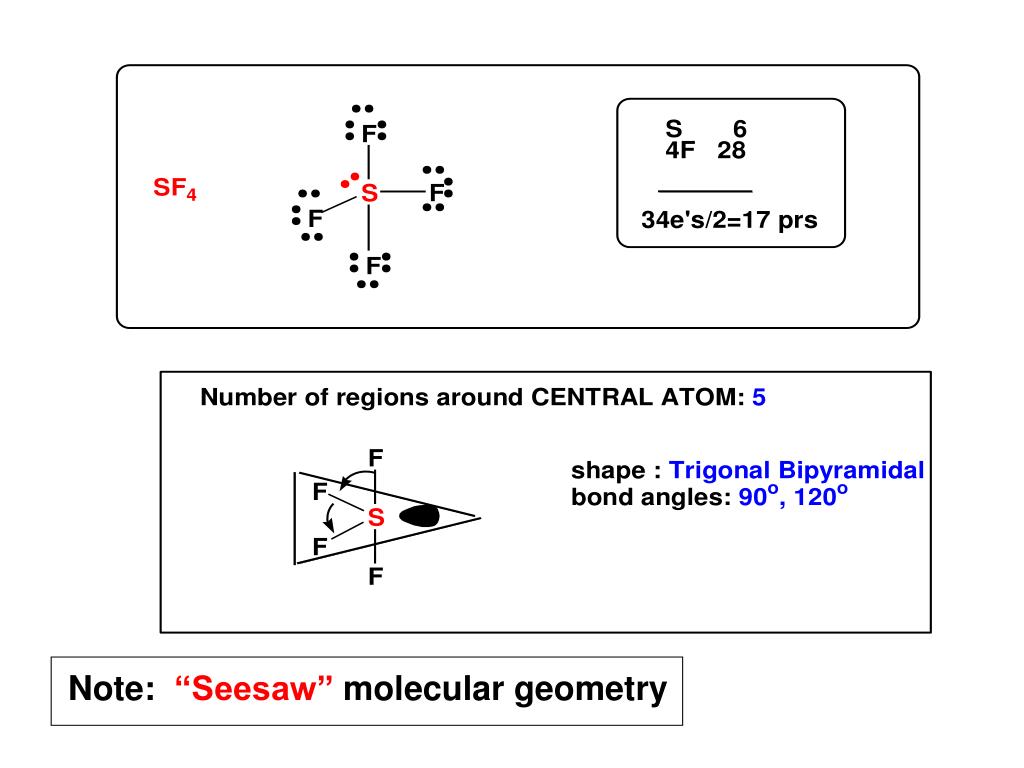
I know this doesn't actually answer your question, but I hope that it explains why your question is flawed. This corresponds well to simple overlap between the p orbitals of sulfur with the s orbital of hydrogen. molecular geometries in this domain: Trigonal bipyramidal Seesaw T-shaped Linear. In larger atoms however, we do not always need to invoke hybridization. This is the Valence-Shell Electron-Pair Repulsion (VSEPR) model. seesaw molecular XeCl4 geometry octahedral electron sp geometry BeF2 polar nonpolar, but contains a sp2 polar covalent bond linear : BCI3 trigonal planar sp3. Hybridization needs to be invoked to rationalize the molecular geometries around small atoms (O,N,C, etc) with the geometry of the atomic orbitals in these atoms. Normally, when all these regions are bonding, the molecule has 120 degree angles between the three atoms making up the 'trigonal' part of the shape and 90 degree angles between the two atoms of the 'bipyramidal' part of the shape in relation to the other atoms. One of the problems here is that instructors (and textbooks), especially at an introductory level will have a tendency to over-generalize the rules for Lewis diagrams and for hybridization. For the seesaw shape, we have 5 regions of electron density (trigonal bipyramidal), consisting of 4 bonding pairs and 1 lone pair. As such, the sp 3d and sp 3d 2 hybridizations are incorrect. As others have mentioned, it has been shown that d-orbitals do not participate in hybridization. Sulfur hexafluoride, for example appears to be a covalent-ionic hybrid.Īt the same time, Lewis drawings of hypervalent species such as sulfate ion are still commonly used in organic chemistry, biochemistry and biology. This means that CO2 molecules are non-polar and will not be very soluble in water (a polar solvent). 
For example, carbon dioxide is a linear molecule. Explanation: The shape of a molecule helps to determine its properties. Seesaw geometry is a form of molecular geometry in which the central atom has one lone pair of electrons and is coupled to four bonding groups. This Wikipedia article does a reasonably good job at looking at the issue: hypervalent molecule.īasically, the even though we can draw Lewis structures of hypervalent compounds, it doesn't mean that they exist as such. Answer: Molecular geometry is used to determine the shapes of molecules. For example, if we want to obtain the Lewis structure of the Sulfate ion. Hypervalency (expanded octets) don't appear to actually exist. It forms a see-saw shape and has a trigonal bipyramidal molecular geometry. The valence shell electron pair repulsion (VSEPR) model focuses on the bonding and nonbonding electron pairs present in the outermost (valence) shell of an atom that connects with two or more other atoms.įundamentally, the VSEPR model theorizes that these regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible.This is a complicated issue, and there are many problems with what you are asking. one electron pair include the pair PdH,- and ClF3, which are T-shaped, and RhB, and SF, which both adopt seesaw geometries.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
